|
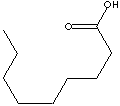
n-NONANOIC ACID |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PRODUCT IDENTIFICATION |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS NO. |
112-05-0 |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| EINECS NO. | 203-931-2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FORMULA | CH3(CH2)7COOH | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MOL WT. | 158.24 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H.S. CODE | 2915.90 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
TOXICITY |
Oral rat LD50: >5 gm/kg | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SYNONYMS | 1-Octanecarboxylic acid; n-Pelargonic Acid; | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Nonanoic Acid; 1-octanecarboxylic Acid; Nonylic Acid; Acide Nonylique Normal; Acide Pelargonique; Hexacid C-9; Octane-1-carboxylic Acid; Pelargic Acid; Pelargon (Russian); | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PRICE |
U$50/kg CFR by sea
for 100kgs |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
CLASSIFICATION |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
PHYSICAL AND CHEMICAL PROPERTIES |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PHYSICAL STATE |
clear to yellowish oily liquid |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MELTING POINT | 11 C | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BOILING POINT |
253 C |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SPECIFIC GRAVITY |
0.907 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SOLUBILITY IN WATER | Insoluble | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pH | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| VAPOR DENSITY | 5.45 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AUTOIGNITION |
405 C |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NFPA RATINGS | Health: 3 Flammability: 1 Reactivity: 0 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
REFRACTIVE INDEX |
1.4322 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FLASH POINT |
114 C |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| STABILITY | Stable under ordinary conditions | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
DESCRIPTION AND APPLICATIONS |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Pelargonic acid is a clear to yellowish oily liquid. It is insoluble in water but soluble in ether, alcohol and organic solvents. The molecules of most natural fatty acids have an even number of carbon chains due to the linkage together by ester units. Analogous compounds of odd numbers carbon chain fatty acids are supplemented synthetically. Pelargonic acid, C-9 odd numbers carbon chain fatty acid, is relatively high cost fatty acid. Pelargonic acid can be prepared by ozonolysis which uses ozone is to cleave the alkene bonds. Eexample of ozonolysis in commerce is the production of odd carbon number carboxylic acids such as azelaic acid and pelargonic acid and simple carboxylic acids such as formic acid and oxalic acid. Pelargonic acid forms esters with alcohols to be used as plasticizers and lubricating oils. It is used in modifying alkyd resins to prevent discolor and to keep flexibility and resistance to aging since saturated pelargonic acid will not be oxidized. Metallic soaps (barium and cadmium) and other inorganic salts used as a stabilizer. It is also used as a chemical intermediate for synthetic flavors, cosmetics, pharmaceuticals and corrosion inhibitors. It is known that C8 - C12 straight and saturated chain fatty acids are capable of removing the waxy cuticle of the broadleaf or weed, resulting in causing the tissue death. They are used as active ingredient of environment friendly and quick effect herbicides. Pelargonic acid is the strongest one. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SALES SPECIFICATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
APPEARANCE |
clear to yellowish oily liquid |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CONTENT OF C9 ACIDS |
98.5% min |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
2-METHYL OCTANOIC ACID |
2.5% max |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
WATER |
0.1% max |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
IODINE VALUE |
0.6 max |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| COLOR |
25 max(Pt/Co scale) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TRANSPORTATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PACKING | 180kgs in drum | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HAZARD CLASS | Not regulated | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| UN NO. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PRICE INDICATION | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| USD38.00/kg CFR by sea for the quantity 180kgs shipment. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GENERAL DESCRIPTION OF CARBOXYLIC ACID | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Carboxylic acid is an organic compound whose molecules contain carboxyl group
and have the condensed chemical formula R-C(=O)-OH in which a carbon atom is
bonded to an oxygen atom by a solid bond and to a hydroxyl group by a single
bond), where R is a hydrogen atom, an alkyl group, or an aryl group. Carboxylic
acids can be synthesized if aldehyde is oxidized. Aldehyde can be obtained by
oxidation of primary alcohol. Accordingly, carboxylic acid can be obtained by
complete oxidation of primary alcohol. A variety of Carboxylic acids are
abundant in nature and many carboxylic acids have their own trivial names.
Examples are shown in table. In substitutive nomenclature, their names are
formed by adding -oic acid' as the suffix to the name of the parent compound.
The first character of carboxylic acid is acidity due to dissociation into H+
cations and RCOO- anions in aqueous solution. The two oxygen atoms are
electronegatively charged and the hydrogen of a carboxyl group can be easily
removed. The presence of electronegative groups next to the carboxylic group
increases the acidity. For example, trichloroacetic acid is a stronger acid than
acetic acid. Carboxylic acid is useful as a parent material to prepare many
chemical derivatives due to the weak acidity of the hydroxyl hydrogen or due to
the difference in electronegativity between carbon and oxygen. The easy
dissociation of the hydroxyl oxygen-hydrogen provide reactions to form an ester
with an alcohol and to form a water-soluble salt with an alkali. Almost infinite
esters are formed through condensation reaction called esterification between
carboxylic acid and alcohol, which produces water. The second
reaction theory is the addition of electrons to the electron-deficient carbon
atom of the carboxyl group. One more theory is decarboxylation (removal of
carbon dioxide form carboxyl group). Carboxylic acids are used to synthesize
acyl halides and acid anhydrides which are generally not target compounds. They
are used as intermediates for the synthesis esters and amides, important
derivatives from carboxylic acid in biochemistry as well as in industrial
fields. There are almost
infinite esters obtained from carboxylic
acids. Esters
are formed by removal of water from an acid and an alcohol. Carboxylic acid
esters are used as in a variety of direct and indirect applications. Lower chain
esters are used as flavouring base materials, plasticizers, solvent carriers and
coupling agents. Higher chain compounds are used as components in metalworking
fluids, surfactants, lubricants, detergents, oiling agents, emulsifiers, wetting
agents textile treatments and emollients, They are also used as intermediates
for the manufacture of a variety of target compounds. The almost infinite esters
provide a wide range of viscosity, specific gravity, vapor pressure, boiling
point, and other physical and chemical properties for the proper application
selections. Amides
are formed from the reaction of a carboxylic acids with an amine.
Carboxylic
acid's reaction to link amino acids is wide in nature to form proteins (amide), the
principal constituents of the protoplasm of all cells. Polyamide is a polymer
containing repeated amide groups such as various kinds of nylon and
polyacrylamides. Carboxylic acid
are in our lives.
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||